Wetenschap
Nieuwe silicium nanodraden kunnen echt tegen de hitte

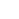
Transmissie-elektronenmicroscoopopname van silicium-28 nanodraad met een laag siliciumdioxide op het oppervlak. Credit:Matthew R. Jones en Muhua Sun/Rice University
Wetenschappers hebben een nieuw materiaal aangetoond dat warmte 150% efficiënter geleidt dan conventionele materialen die worden gebruikt in geavanceerde chiptechnologieën.
Het apparaat - een ultradunne silicium nanodraad - zou kleinere, snellere micro-elektronica mogelijk maken met een warmteoverdrachtsefficiëntie die de huidige technologieën overtreft. Elektronische apparaten aangedreven door microchips die warmte efficiënt afvoeren, zouden op hun beurt minder energie verbruiken - een verbetering die zou kunnen helpen het energieverbruik te verminderen dat wordt geproduceerd door de verbranding van koolstofrijke fossiele brandstoffen die hebben bijgedragen aan de opwarming van de aarde.
"Door de natuurlijke beperkingen van silicium in zijn vermogen om warmte te geleiden te overwinnen, pakt onze ontdekking een hindernis in microchip-engineering aan", zegt Junqiao Wu, de wetenschapper die de Physical Review Letters leidde. studie die het nieuwe apparaat rapporteert. Wu is een faculteitswetenschapper in de Materials Sciences Division en hoogleraar materiaalwetenschappen en engineering aan UC Berkeley.
De langzame stroom van warmte door silicium
Onze elektronica is relatief betaalbaar omdat silicium - het materiaal bij uitstek voor computerchips - goedkoop en overvloedig is. Maar hoewel silicium een goede geleider van elektriciteit is, is het geen goede warmtegeleider wanneer het wordt verkleind tot zeer kleine afmetingen - en als het gaat om snel computergebruik, vormt dat een groot probleem voor kleine microchips.
Binnen elke microchip bevinden zich tientallen miljarden siliciumtransistoren die de stroom van elektronen in en uit geheugencellen sturen, en databits coderen als enen en nullen, de binaire taal van computers. Tussen deze hardwerkende transistors lopen elektrische stromen en deze stromen genereren onvermijdelijk warmte.
Warmte stroomt van nature van een heet object naar een koud object. Maar warmtestroom wordt lastig in silicium.
In zijn natuurlijke vorm bestaat silicium uit drie verschillende isotopen - vormen van een chemisch element met een gelijk aantal protonen maar een verschillend aantal neutronen (vandaar verschillende massa) in hun kernen.
Ongeveer 92% van silicium bestaat uit de isotoop silicium-28, die 14 protonen en 14 neutronen heeft; ongeveer 5% is silicium-29, met een gewicht van 14 protonen en 15 neutronen; en slechts 3% is silicium-30, een relatief zwaargewicht met 14 protonen en 16 neutronen, legt co-auteur Joel Ager uit, die titels heeft van senior wetenschapper in de Materials Sciences Division van Berkeley Lab en adjunct-professor materiaalwetenschappen en engineering aan UC Berkeley.
Als fononen, de golven van atomaire trillingen die warmte transporteren, zich een weg banen door de kristallijne structuur van silicium, verandert hun richting wanneer ze tegen silicium-29 of silicium-30 botsen, waarvan de verschillende atoommassa's de fononen "verwarren", waardoor ze worden vertraagd.
"The phonons eventually get the idea and find their way to the cold end to cool the silicon material," but this indirect path allows waste heat to build up, which in turn slows your computer down, too, Ager said.
A big step toward faster, denser microelectronics
For many decades, researchers theorized that chips made of pure silicon-28 would overcome silicon's thermal conductivity limit, and therefore improve the processing speeds of smaller, denser microelectronics.
But purifying silicon down to a single isotope requires intense levels of energy which few facilities can supply—and even fewer specialize in manufacturing market-ready isotopes, Ager said.
Fortunately, an international project from the early 2000s enabled Ager and leading semiconductor materials expert Eugene Haller to procure silicon tetrafluoride gas—the starting material for isotopically purified silicon—from a former Soviet-era isotope manufacturing plant.
This led to a series of pioneering experiments, including a 2006 study published in Nature , whereby Ager and Haller fashioned silicon-28 into single crystals, which they used to demonstrate quantum memory storing information as quantum bits or qubits, units of data stored simultaneously as a one and a zero in an electron's spin.
Subsequently, semiconducting thin films and single crystals made with Ager's and Haller's silicon isotope material were shown to have a 10% higher thermal conductivity than natural silicon—an improvement, but from the computer industry's point of view, probably not enough to justify spending a thousand times more money to build a computer from isotopically pure silicon, Ager said.
But Ager knew that the silicon isotope materials were of scientific importance beyond quantum computing. So he kept what remained in a safe place at Berkeley Lab, just in case other scientists might need it, because few people have the resources to make or even purchase isotopically pure silicon, he reasoned.
A path toward cooler tech with silicon-28
About three years ago, Wu and his graduate student Penghong Ci were trying to come up with new ways to improve the heat transfer rate in silicon chips.
One strategy to make more efficient transistors involves using a type of nanowire called a Gate-All-Around Field Effect Transistor. In these devices, silicon nanowires are stacked to conduct electricity, and heat is generated simultaneously, Wu explained. "And if the heat generated is not extracted out quickly, the device would stop working, akin to a fire alarm blaring in a tall building without an evacuation map," he said.
But heat transport is even worse in silicon nanowires, because their rough surfaces—scars from chemical processing—scatter or "confuse" the phonons even more, he explained.
"And then one day we wondered, 'What would happen if we made a nanowire from isotopically pure silicon-28?'" Wu said.
Silicon isotopes are not something one can easily buy on the open market, and word had it that Ager still had some silicon isotope crystals in storage at Berkeley Lab—not a lot, but still enough to share "if someone has a great idea about how to use it," Ager said. "And Junqiao's new study was such a case."
A surprising big reveal with nano tests
"We're really fortunate that Joel happened to have the isotopically enriched silicon material ready to use for the study," Wu said.
Using Ager's silicon isotope materials, the Wu team tested the thermal conductivity in bulk 1-millimeter-size silicon-28 crystals versus natural silicon—and again, their experiment confirmed what Ager and his collaborators discovered years ago—that bulk silicon-28 conducts heat only 10% better than natural silicon.
Now for the nano test. Using a technique called electroless etching, Ci made natural silicon and silicon-28 nanowires just 90 nanometers (billionths of a meter) in diameter—about a thousand times thinner than a single strand of human hair.
To measure the thermal conductivity, Ci suspended each nanowire between two microheater pads outfitted with platinum electrodes and thermometers, and then applied an electrical current to the electrode to generate heat on one pad that flows to the other pad via the nanowire.
"We expected to see only an incremental benefit—something like 20%—of using isotopically pure material for nanowire heat conduction," Wu said.
But Ci's measurements astonished them all. The Si-28 nanowires conducted heat not 10% or even 20%, but 150% better than natural silicon nanowires with the same diameter and surface roughness.
This defied everything that they had expected to see, Wu said. A nanowire's rough surface typically slows phonons down. So what was going on?
High-resolution TEM (transmission electron microscopy) images of the material captured by Matthew R. Jones and Muhua Sun at Rice University uncovered the first clue:a glass-like layer of silicon dioxide on the silicon-28 nanowire surface.
Computational simulation experiments at the University of Massachusetts Amherst led by Zlatan Aksamija, a leading expert on the thermal conductivity of nanowires, revealed that the absence of isotope "defects"—silicon-29 and silicon-30—prevented phonons from escaping to the surface, where the silicon dioxide layer would drastically slow down the phonons. This in turn kept phonons on track along the direction of heat flow—and therefore less "confused"—inside the silicon-28 nanowire's "core." (Aksamija is currently an associate professor of materials science and engineering at the University of Utah.)
"This was really unexpected. To discover that two separate phonon-blocking mechanisms—the surface versus the isotopes, which were previously believed to be independent of each other—now work synergistically to our benefit in heat conduction is very surprising but also very gratifying," Wu said.
"Junqiao and the team discovered a new physical phenomenon," Ager said. "This is a real triumph for curiosity-driven science. It's quite exciting."
Wu said that the team next plans to take their discovery to the next step:by investigating how to "control, rather than merely measure, heat conduction in these materials." + Verder verkennen
Thermoelectric silicon material reaches record-low thermal conductivity
 Zelfherstellend beton kan de levensduur van constructies verlengen
Zelfherstellend beton kan de levensduur van constructies verlengen Video:Waarom schelpen sterker zijn dan krijt
Video:Waarom schelpen sterker zijn dan krijt Warmte omzetten in kracht met efficiënt organisch thermo-elektrisch materiaal
Warmte omzetten in kracht met efficiënt organisch thermo-elektrisch materiaal Kameleonmaterialen:de oorsprong van kleurvariatie in laagdimensionale perovskieten
Kameleonmaterialen:de oorsprong van kleurvariatie in laagdimensionale perovskieten Onderzoekers ontwikkelen een methode om miljoenen potentiële zelfgeproduceerde kandidaat-geneesmiddelen in één keer te onderzoeken
Onderzoekers ontwikkelen een methode om miljoenen potentiële zelfgeproduceerde kandidaat-geneesmiddelen in één keer te onderzoeken
 Arseen in grondwater? Virginia kolenas zaak voor de rechtbank
Arseen in grondwater? Virginia kolenas zaak voor de rechtbank Experts ontwikkelen kunstmatige intelligentie om waterkwaliteit beter te monitoren
Experts ontwikkelen kunstmatige intelligentie om waterkwaliteit beter te monitoren Wat Facebook ons kan vertellen over voedingskeuzes
Wat Facebook ons kan vertellen over voedingskeuzes Welke dieren tonen commensalisme in het regenwoud?
Welke dieren tonen commensalisme in het regenwoud?  De positieve kanten van klimaatverandering? Onderzoek toont aan dat landbouw, economische mogelijkheden
De positieve kanten van klimaatverandering? Onderzoek toont aan dat landbouw, economische mogelijkheden
Hoofdlijnen
- Hoe een utility-functie af te leiden
- Zooplankton Vs. fytoplankton
- Driedimensionale fossiele algen van meer dan 541 miljoen jaar oud onthullen modern ogende voorouders van het plantenrijk
- Death Valley wordt elke 10 jaar gevuld met wilde bloemen - het gebeurt nu!
- Wetenschappers rapporteren chemische basis van de celdelingstimer, een mogelijk doelwit tegen kanker
- Wat betekent ion?
- Onderzoekers laten zien hoe voedselkeuze voor insecten kan worden gemanipuleerd
- Twee soorten Cilia in een Paramecium
- Sluwe kraaien weten wat er nodig is om een goed stuk gereedschap te maken
- Nieuwe nanodeeltjes verzwakken de afweer van tumorcellen, sla dan toe met chemotherapiemedicijn
- 2D-materialen combineren, gepolariseerd raken en aanleiding geven tot fotovoltaïsch effect
- De mogelijkheden van de nanowereld benutten

- Apparaten van de volgende generatie krijgen een boost van grafeenonderzoek

- Gevaarlijke stoffen uit de lucht halen

 Kernbrandstof verwijderd uit kreupele fabriek in Japan
Kernbrandstof verwijderd uit kreupele fabriek in Japan Giftige bijproducten van Agent Orange blijven het milieu in Vietnam vervuilen, studie zegt:
Giftige bijproducten van Agent Orange blijven het milieu in Vietnam vervuilen, studie zegt: Verouderde verontreinigende stoffen gevonden in trekkende sterns in het gebied van de Grote Meren
Verouderde verontreinigende stoffen gevonden in trekkende sterns in het gebied van de Grote Meren Nieuwe mysteries omringen het volgende flyby-doel van New Horizons
Nieuwe mysteries omringen het volgende flyby-doel van New Horizons Belangrijke feiten over het ecosysteem Open Oceaan
Belangrijke feiten over het ecosysteem Open Oceaan Hoe maak je een zelfgemaakte thermosfles voor een Science Fair-project
Hoe maak je een zelfgemaakte thermosfles voor een Science Fair-project  De eerste mensen in Tasmanië moeten spectaculaire aurora's hebben gezien
De eerste mensen in Tasmanië moeten spectaculaire aurora's hebben gezien Wereldprimeur voor het lezen van digitaal gecodeerde synthetische moleculen
Wereldprimeur voor het lezen van digitaal gecodeerde synthetische moleculen
- Elektronica
- Biologie
- Zonsverduistering
- Wiskunde
- French | Italian | Spanish | Portuguese | Swedish | German | Dutch | Danish | Norway |

-
Wetenschap © https://nl.scienceaq.com