Wetenschap
Hoe cellen hun grenzen beheren
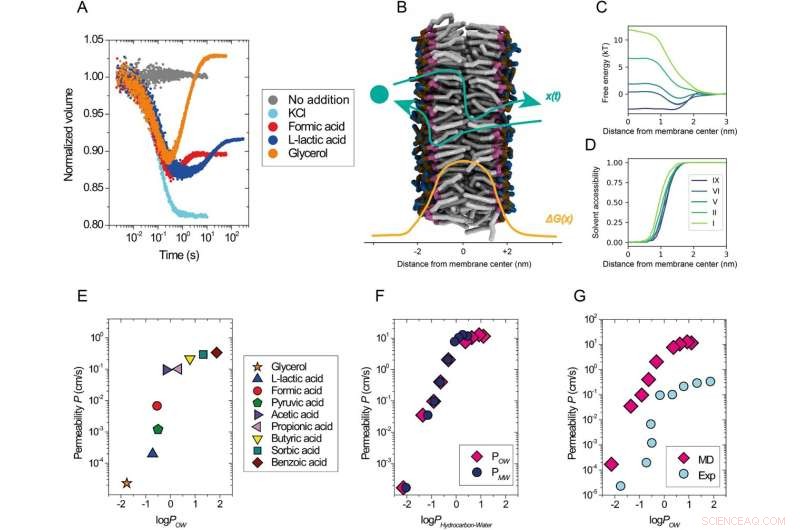
Permeabiliteit van opgeloste stoffen als functie van hun lipofiliciteit. Een overzicht van experimentele analyse. Kinetische gegevens verkregen met de zelfdovende calceïne-assay met blaasjes die zijn samengesteld uit DOPC gemengd met buffer (grijs) of osmotisch geshockeerd met 52,5 mM KCl (cyaan), 50 mM natriumformiaat (rood), 50 mM natrium L-lactaat (blauw) of 120 mM glycerol (oranje) bij 20 °C. B Schematische beschrijving van het permeatieproces, x(t), door een lipidemembraan met een voorbeeld van een vrij energieprofiel, ΔG(x) (lipidestaarten, grijs; glycerolgroep, paars; fosfaatgroep, oker; en cholinegroep, blauw; watermoleculen worden niet getoond). C Geselecteerde vrije-energieprofielen uit simulaties van opgeloste stoffen met variërende hydrofobiciteitsniveaus (I meest hydrofiel, IX meest hydrofoob) die door een DOPC-lipidemembraan doordringen als een functie van de afstand vanaf het dubbellaagscentrum langs het membraan. Slechts de helft van het gehele symmetrische permeatieprofiel wordt getoond. D Oplosmiddeltoegankelijkheidsprofielen van de doordringende opgeloste stoffen langs het permeatiepad. Opgeloste stoffen interageren met oplosmiddelmoleculen, zelfs diep in het membraanstaartgebied (x < 1.0 nm). E–G Permeabiliteitscoëfficiënten voor DOPC-membranen uit experimenten bij 20 °C (E, G) en simulaties (F, G) uitgezet tegen de logaritme van octanol/water (logPOW ) en membraan/water-verdelingscoëfficiënt (logPMW ) van de opgeloste stoffen. Opgeloste stoffen van de hydrofobe niveaus I (logPOW = −2.14) tot IX (logPOW = 1.1) werden gebruikt in de simulaties. LogP is log10 (partitiecoëfficiënt), waarbij P verwijst naar de evenwichtsverdeling van een molecuul tussen de hydrofobe en hydrofiele fase van twee niet-mengbare oplosmiddelen. Het experimentele logboekPOW waarden voor zwakke zuren en glycerol (E, G) zijn ontleend aan de PubChem-database (//pubchem.ncbi.nlm.nih.gov/). De gesimuleerde logPOW waarden (F, G) zijn overgenomen uit werk25; logPMW waarden (F) werden verkregen uit de huidige simulaties. De verdelingscoëfficiënten zijn gerelateerd aan het vrije energieverschil tussen de respectievelijke fasen. In het geval van PMW , dit is het verschil tussen het membraancentrum en de oplosmiddelfase. De waarden van de vrije energie zijn weergegeven in (C). Krediet:Natuurcommunicatie (2022). DOI:10.1038/s41467-022-29272-x
Bacteriën, schimmels en gisten zijn erg goed in het uitscheiden van nuttige stoffen zoals zwakke zuren. One way in which they do this is through passive diffusion of molecules across the cell membrane. At the same time, cells need to prevent leakage of numerous small molecules. Yeast cells, for instance, can live in hostile environments thanks to a very robust and relatively impermeable membrane system. Biochemists at the University of Groningen have studied how the composition of the membrane affects passive diffusion and the robustness of the cell membrane. Their results, which were published in Nature Communications on March 25, could help the biotech industry to optimize microbial production of useful molecules and help in drug design.
Border control is very important to cells. Their membranes separate the inner and outer environments, which are quite different. To absorb useful compounds, such as nutrients, or to excrete waste, cells can use selective transport systems. However, some transport across the membrane takes place by passive diffusion. This is a non-selective process that will let some molecules go in or out, depending on their size and hydrophobicity, for example. Active transporters have been studied extensively; however, our knowledge of passive diffusion through the membrane is still very incomplete.
Synthetic vesicles
This is a problem for the biotechnology industry, which uses cells as factories to produce a myriad of useful substances and that needs these worker cells to survive under harsh conditions, for example in an environment with a high alcohol or weak acid concentration. Bert Poolman, Professor of Biochemistry at the University of Groningen, was approached by a biotech company that was interested in producing lactic acid in bacteria. They wanted to know more about passive diffusion. This fitted in nicely with another project that Poolman is working on. "We are highly interested in these passive transport processes because of our involvement in a project to build a synthetic cell," says Poolman. "If you can use passive diffusion instead of an active transport system, you need fewer parts to construct such a cell."
So, he combined both questions in a research project. "We started out with a systematic study of what causes the differences in permeability of yeast membranes and bacterial membranes," says Poolman. His team created synthetic vesicles that were made up of three to four different lipids. Ergosterol or cholesterol was added to the membranes to affect their fluidity and rigidity. A range of small molecules was tested using this system and the results from these experiments guided molecular dynamic simulations of diffusion through membranes. The in-silico studies, supervised by Professor Siewert-Jan Marrink, provided a deeper insight into the molecular mechanism of diffusion.
Tweaking
The fatty acid tails of the lipids turned out to be most important in determining the properties of membranes, whereas the hydrophilic head groups had little effect on the permeability. The length of the tails also mattered. "And saturated tails, with no double carbon bonds, are stiffer than unsaturated ones. Hydrophobic interactions cause a close packing of these tails, resulting in a gel phase that is not very penetrable," explains Poolman. Sterols increase the fluidity but in the case of yeast, which uses ergosterol, the permeability remains low. "Thus, by tweaking the saturation of the fatty acids and the type and amount of sterol in the membrane, we can modify the permeability of the plasma membrane of yeast and bacterial cells."
Poolman and his colleagues have, therefore, defined a number of variables that alter the permeability of membranes for different classes of compounds. This information can be used by companies that use yeasts or bacteria as cell factories. "However, our results cannot be directly applied to those cells," says Poolman. "Real membranes contain hundreds of different lipids and the composition can vary between different locations in the membrane. In addition, these cell membranes contain all kinds of proteins. If you make changes in, for example, the lipid composition of the membrane, a lot can go wrong and the function of a membrane protein can be affected."
Drug design
The increased understanding of the physical processes that affect permeability can help companies to understand why certain cells are better for specific processes than others. "The usual way to tweak strains is by directed evolution. Our results will help companies to better understand the results of those optimizations and guide their cell engineering efforts."
Another application is the design of drugs that act inside cells. "Pharmaceutical companies use a set of empirically established rules to optimize drugs for action inside cells, based on parameters such as size or polarity. Our study highlights the importance of the membrane composition of the targeted cells and this could help in drug design."
 Zoete wetenschap:glucosestroop aan het werk zetten op de oorsprong van de aarde
Zoete wetenschap:glucosestroop aan het werk zetten op de oorsprong van de aarde NASA lanceert geavanceerde laser om het veranderende ijs van de aarde te meten
NASA lanceert geavanceerde laser om het veranderende ijs van de aarde te meten Van jeans tot vezels tot kledinglabels - nieuwe recycling voor duurzamere mode
Van jeans tot vezels tot kledinglabels - nieuwe recycling voor duurzamere mode Hoeveel protozoa zitten er in het water dat we drinken?
Hoeveel protozoa zitten er in het water dat we drinken? Verder gaan dan de groene revolutie in het nieuwe hongertijdperk van Afrika
Verder gaan dan de groene revolutie in het nieuwe hongertijdperk van Afrika
Hoofdlijnen
- Ondergewaardeerde microben krijgen nu de eer voor het behouden van twee banen in de bodem
- Wat zijn de verschillen tussen een centraal zenuwstelsel en een centraal zenuwstelsel?
- Hoe een molecuul met twee gezichten problematische genen tot zwijgen kan brengen
- Chinese fossiele eieren tonen achteruitgang van dinosauriërs voordat ze uitstierven
- Zenuwen controleren de bacteriële gemeenschap van het lichaam
- Hoe Dopamine werkt
- Hoe bacteriën werken
- Hoe transcriptiefactoren clusters van specifieke DNA-sequenties vinden en herkennen
- Graafwespen en hun chemie
- Het leefgebied van de panda krimpt, steeds meer gefragmenteerd worden

- Zijn getrouwde mensen gelukkiger dan alleenstaanden?

- Studie suggereert nieuwe doelen voor het verbeteren van het sojaoliegehalte

- Onderzoekers verwachten dat nieuwe vispassage de trekvispopulaties zal herstellen

- Wetenschappers speuren naar een ongrijpbare plantenziekteverwekker in Mexico

 Een methode om canonieke fasemetingen uit te voeren met behulp van kwantumfeedback
Een methode om canonieke fasemetingen uit te voeren met behulp van kwantumfeedback Hoe seks en gender invloed hebben op hoe we stemmen
Hoe seks en gender invloed hebben op hoe we stemmen Nieuwe studie vindt een economische impact van $ 1 miljoen per mijl van TVA-reservoirs
Nieuwe studie vindt een economische impact van $ 1 miljoen per mijl van TVA-reservoirs Zuckerberg zegt dat de passiviteit van de Amerikaanse regering het verspreiden van nepnieuws mogelijk maakte
Zuckerberg zegt dat de passiviteit van de Amerikaanse regering het verspreiden van nepnieuws mogelijk maakte Nieuwe wiskundige methode laat zien hoe klimaatverandering leidde tot de ondergang van de oude beschaving
Nieuwe wiskundige methode laat zien hoe klimaatverandering leidde tot de ondergang van de oude beschaving Studie onthult oorsprong van beweging van de oceaan in de Straat van Florida
Studie onthult oorsprong van beweging van de oceaan in de Straat van Florida Hybride nanomaterialen barsten van de potentie
Hybride nanomaterialen barsten van de potentie Afbeelding:MetOp-C-payloadmodule
Afbeelding:MetOp-C-payloadmodule
- Elektronica
- Biologie
- Zonsverduistering
- Wiskunde
- French | Italian | Spanish | Portuguese | Swedish | German | Dutch | Danish | Norway |

-
Wetenschap © https://nl.scienceaq.com